Rare Immunology News
Advertisement
Spotlight On
Activated PI3K Delta Syndrome (APDS)
Activated PI3K Delta Syndrome (APDS) is a rare, genetic primary immunodeficiency
Prevalence
< 1 / 1,000,000
Prevalence
Age of Onset

ICD-10
D81.8
Inheritance
Autosomal dominant

Autosomal recessive

Mitochondrial/Multigenic

X-linked dominant

X-linked recessive

Rare View
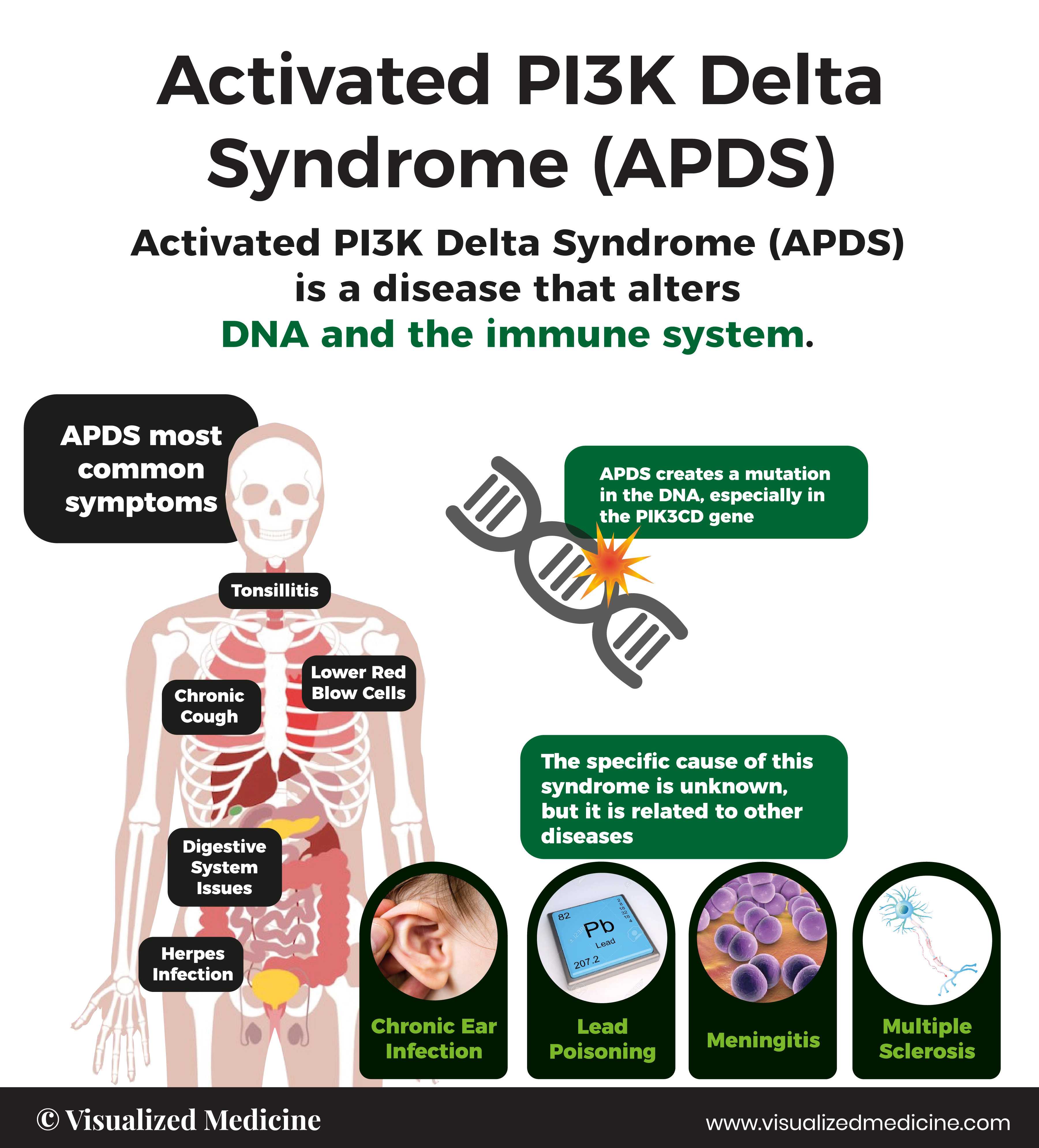
Activated PI3K delta syndrome, known as APDS (previously known as PASLI* Disease) is a rare primary immunodeficiency, first discovered in 2013. APDS is caused by genetic variants in either one of two genes known as PIK3CD or PIK3R1, which encode proteins that are vital to the normal development and function of immune cells. Signs and symptoms of APDS start in childhood, and patients are vulnerable to repeat infections and immune dysregulation such as lymphadenopathy, splenomegaly, autoimmune cytopenias, and even lymphoma.†
5 Facts you should know
FACT
APDS a Primary Immunodeficiency, was only characterized in 2013 though shares many features of other immune disorders, which means APDS patients may have been previously misdiagnosed with other conditions.
FACT
Signs and symptoms of APDS start in childhood, and patients are vulnerable to repeat infections and immune dysregulation such as sinusitis, severe respiratory tract infections, recurrent herpes or EBV/CMV, lymphadenopathy and autoimmune cytopenias.
FACT
Immunologists may mistake APDS for combined immunodeficiency (CID) or common variable immune deficiency (CVID) or hyper IgM syndrome (HIGM).
FACT
Genetic testing is the only way to definitively diagnose APDS and other primary immunodeficiencies.
FACT
Management of APDS is symptomatic – antibiotics, antivirals or IVIG may be used to treat or prevent recurrent infections immunodeficiencies.
Interest Over Time
Google searches
Common Signs & Symptoms
Bronchiectasis
Permanent enlargement of the airways of the lungs
Decreased circulating IgG2 level
Decreased proportion of class-switched memory B cells
Decreased specific pneumococcal antibody level
Increased circulating IgM level
Increased proportion of transitional B cells
Lymphadenopathy
Recurrent ear infections
† All About APDS https://allaboutapds.com